
РЕЗОЛЮЦІЯ: Громадського обговорення навчальної програми статевого виховання
ЧОМУ ФОНД ОЛЕНИ ПІНЧУК І МОЗ УКРАЇНИ ПРОПАГУЮТЬ "СЕКСУАЛЬНІ УРОКИ"
ЕКЗИСТЕНЦІЙНО-ПСИХОЛОГІЧНІ ОСНОВИ ПОРУШЕННЯ СТАТЕВОЇ ІДЕНТИЧНОСТІ ПІДЛІТКІВ
Батьківський, громадянський рух в Україні закликає МОН зупинити тотальну сексуалізацію дітей і підлітків
Відкрите звернення Міністру освіти й науки України - Гриневич Лілії Михайлівні
Представництво українського жіноцтва в ООН: низький рівень культури спілкування в соціальних мережах
Гендерна антидискримінаційна експертиза може зробити нас моральними рабами
ЛІВИЙ МАРКСИЗМ У НОВИХ ПІДРУЧНИКАХ ДЛЯ ШКОЛЯРІВ
ВІДКРИТА ЗАЯВА на підтримку позиції Ганни Турчинової та права кожної людини на свободу думки, світогляду та вираження поглядів
- Гідрологія і Гідрометрія
- Господарське право
- Економіка будівництва
- Економіка природокористування
- Економічна теорія
- Земельне право
- Історія України
- Кримінально виконавче право
- Медична радіологія
- Методи аналізу
- Міжнародне приватне право
- Міжнародний маркетинг
- Основи екології
- Предмет Політологія
- Соціальне страхування
- Технічні засоби організації дорожнього руху
- Товарознавство продовольчих товарів
Тлумачний словник
Авто
Автоматизація
Архітектура
Астрономія
Аудит
Біологія
Будівництво
Бухгалтерія
Винахідництво
Виробництво
Військова справа
Генетика
Географія
Геологія
Господарство
Держава
Дім
Екологія
Економетрика
Економіка
Електроніка
Журналістика та ЗМІ
Зв'язок
Іноземні мови
Інформатика
Історія
Комп'ютери
Креслення
Кулінарія
Культура
Лексикологія
Література
Логіка
Маркетинг
Математика
Машинобудування
Медицина
Менеджмент
Метали і Зварювання
Механіка
Мистецтво
Музика
Населення
Освіта
Охорона безпеки життя
Охорона Праці
Педагогіка
Політика
Право
Програмування
Промисловість
Психологія
Радіо
Регилия
Соціологія
Спорт
Стандартизація
Технології
Торгівля
Туризм
Фізика
Фізіологія
Філософія
Фінанси
Хімія
Юриспунденкция
A case of transformation of heat into work
When the piston moves in the opposite direction, steam enters the cylinder B and used steam is forced out at A. With this simple arrangement, the steam would leave the cylinder on exhaust at a temperature nearly as high as that at which it entered it. A considerable quantity of heat would thus be carried to the condenser or the outside air and lost so far as useful work is concerned. In order to prevent this waste as far as possible, an automatic cutoff is provided. When the piston has moved through about one fourth of its stroke, this slide valve automatically cuts off the supply of steam.
After this cutting off of the steam from the steam chest the steam that has already entered the cylinder expands and pushes the piston forward, through the remainder of the stroke. During this expansion, the piston is doing work, the pressure of the steam is being reduced, and the temperature of the steam is lowered. The heat contained in the steam is thus converted into useful work.
When the piston has reached the end of this stroke, the slide valve opens A and connects В to the steam chest. Live steam is now again admitted to the cylinder behind the piston and it pushes the piston toward the left. The dead steam in front of the piston is forced through A. When, as before, the piston has made about one fourth of its stroke, the slide valve closes B, and the steam behind the piston expands until the piston has reached the end of its stroke. The cycle is then repeated.
The pressure of the steam in the boiler is regulated by means of a pop valve, which allows the steam to escape when the pressure exceeds a certain value.
Gas Engine. - Gas engines and gasoline engines operate on the same principle. In each case the energy is derived from the explosion of a mixture of air and gas or gasoline vapor. The gasoline engine is now the commonest type of engine. It is used to drive motorcars, motorboats, tractors, etc.
Work Done by a Gas Expanding at Constant Pressure. -Let a volume of gas be enclosed behind a piston which is airtight and which moves without friction. Let the pressure acting on the piston be denoted by p and the area of the piston by S. The total force acting on the piston pushing it backward is
F=pS
If, now, the gas in the cylinder is heated, it may be allowed to expand without any change in its pressure, and the piston moves back through a distance x. Work done on piston = force X distance = p X S X x
Now S X x = increase of volume of the gas during expansion.
Hence
Work = p X change in volume
It is convenient to represent the work done by the gas by plotting the pressure of the gas on the vertical axis and the volume of the gas on the horizontal axis. In this case, the pressure is constant for all volumes. Hence, AB represents the relation between the volume and the pressure. If V1 denotes the original volume and V2 the final volume, the length of the line ab represents the change in volume during expansion. The product of the change in volume and the pressure is represented by the rectangle ABba. This area stands for the work done by the gas during its expansion; and since this work is equal to the heat supplied to the gas during expansion, this rectangle also represents the heat taken in by the gas during its expansion.
Work Done by a Gas Expanding at Variable Pressure. -If the gas expands under a variable pressure, the work which it performs may be represented by a diagram similar to that representing the work done by a gas expanding at constant pressure. In this case the line AB (Figure 35), instead of being horizontal as in the preceding case, slopes toward the horizontal axis along which the volumes are plotted. Nevertheless, the area under AB will represent the work done by the gas as it expands with changing pressure. It is possible to construct a rectangle having the same base as the figure ABba and the same area. The height of this rectangle would be the average pressure of the gas during its expansion. If the varying pressure acting on the piston has been replaced by a constant pressure equal to the average pressure, the work done by the gas will remain unchanged. The average pressure may then be defined as the constant pressure by which a varying pressure may be replaced without changing the amount of work done on the piston for the same stroke.
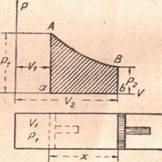
Figure 35 - Work done by a gas at variable pressure
| <== попередня сторінка | | | наступна сторінка ==> |
| | | Equals average pressure times change of volume |
|
Не знайшли потрібну інформацію? Скористайтесь пошуком google: |
© studopedia.com.ua При використанні або копіюванні матеріалів пряме посилання на сайт обов'язкове. |